Vitamin B12 (cobalamin), the only cobalt containing vitamin, is normally obtained from the diet. Upon ingestion the vitamin B12 is released from food in the stomach. Stomach acids and pepsin release protein-bound vitamin B12 from the food, at which time it is bound by a protein called haptocorrin (HC), which is secreted in saliva, forming an Hpatocorin-B12 complex. Vitamin B12 is susceptible to damage from stomach acid, and it is thought that HC protects the cobalamin from damage whilst in the stomach. Release of Gastric acid is dependent upon the release of histamine as part of the cephalic phase of digestion. Production of histamine, requires active vitamin B6. Histamine is required for production of gastric acid, and that then starts the process of gastric emptying from the stomach. Lack of histamine can result in Gastro-Eosophagial Disease (GERD). Treatment often incorrectly uses PPIs.
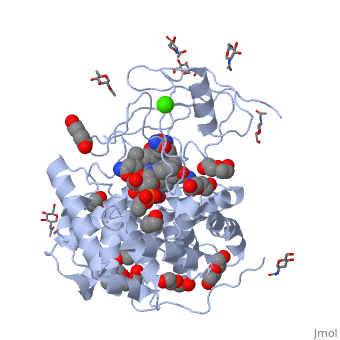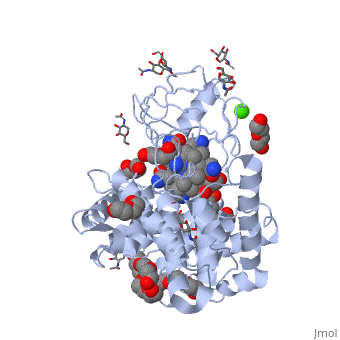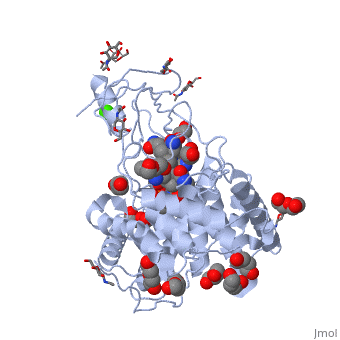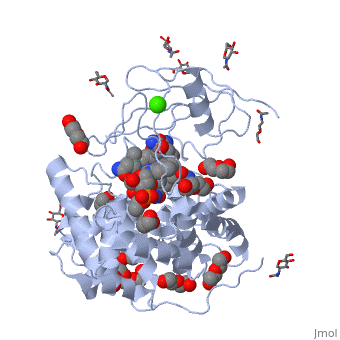
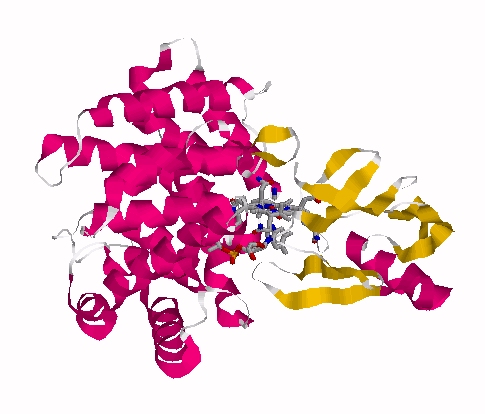
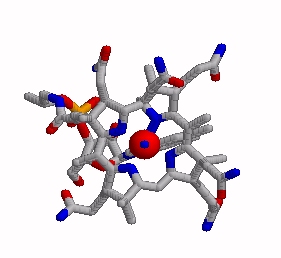
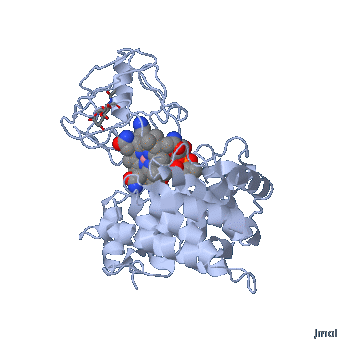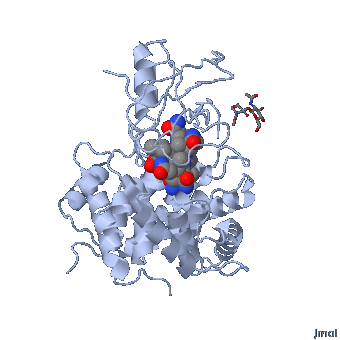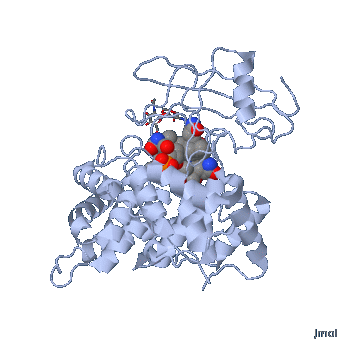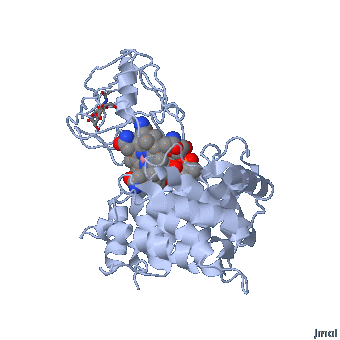
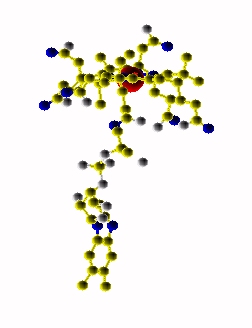
Haptocorrin (HC) bound vitamin B12
Top Jmol revolving Middle Raswin HC-CN-Cbl
Bottom Left CN-Cbl top Right CN-Cbl side
Furger etal, 2003
The haptocorrin-bound vitamin B12 (Hc-B12 complex), travels down into the small intestine where the haptocorrin (HC) is then broken down by intestinal enzymes such as trypsin and chymotrypsin. The protein-free vitamin B12 is then bound by another vitamin B12 binding protein called Intrinsic Factor (IF). Production and secretion of Intrinsic Factor in humans occurs in the parietal cells in the stomach, which are the same cells that secrete gastric acid. Hence if the secretion is blocked or reduced with drugs such as PPIs this step is also reduced, and poor uptake of Vitamin B12 occurs. Further in gastric by-pass surgery, lack of binding of B12 to IF eventually causes B12 deficiency (see section). Vitamin B12 is the largest of the water soluble vitamins, and as such cannot cross the gut wall by simple diffusion, but must be transported across the intestinal epithelial cells and then released into the draining intestinal lymphatics and subsequently released into the circulation. In order for this to occur the Vitamin B12 -IF complex must first bind to a receptor on the wall of the small intestine (Cubilin) (Alpers and Russell-Jones, 2013 Alsenz et al, 2000; Gordon et al, 1997; Wen et al, 2000). The number of receptors on the wall is rather small and hence this is the major limitation to oral delivery of vitamin B12 . Thus, once the intrinsic factor is saturated with vitamin B12 and once the receptors on the gut wall are saturated no further specific uptake of vitamin B12 is possible. Thus, the maximal specific uptake of vitamin B12 per feed is 3-6 ug in humans. This is the reason why it is extremely hard to over-come vitamin B12 deficiency via oral feeding. Basically the amount that crosses the gut wall, which is bound to the specific transporter Transcobalamin (TC), is very, very small.
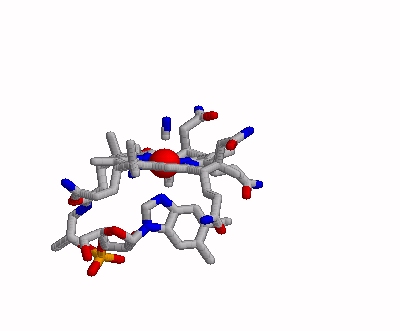
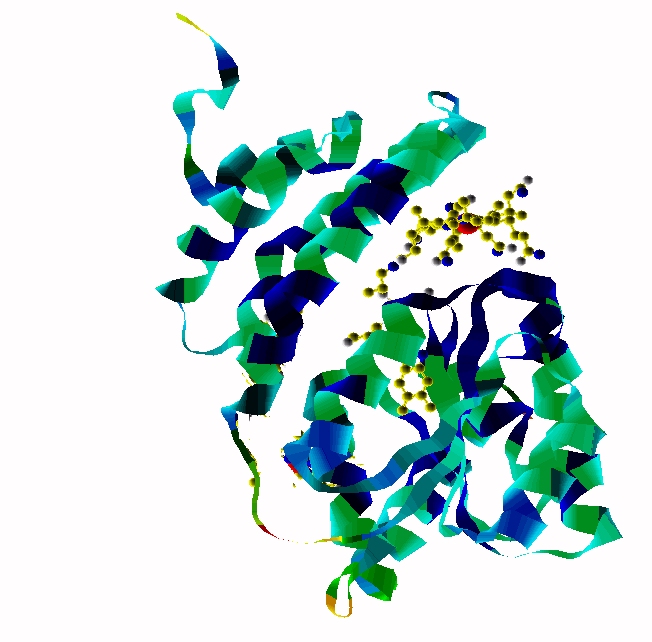
Intrinsic Factor bound vitamin B12
The IF-vitamin B12 complex is internalized by the intestinal epithelial cell and the IF is degraded within the cell and another vitamin B12 binding protein, transcobalamin (formerly called transcobalamin II (TC)) binds to the vitamin B12 .
Transcobalamin-bound vitamin B12
Transcobalamin-bound vitamin B12 is released from the intestinal epithelial cells and travels via the mesenteric lymph vessels until it finally reaches the circulation. During binding of vitamin B12 by transcobalamin (TC), the transcobalamin undergoes a shift in its structure, and is named Holotranscobalamin, and can be distinguished from free transcobalamin (apotranscobalamin) using antibodies. This transcobalamin-B12 has been misnamed "active B12" by several authors, but technically it is the form that is available for uptake into cells. Uptake into cells relies upon the cell having a receptor that recognizes Holotranscobalamin (Holo TC), initially known as the TC-Receptor, but also called CD320. Cobalamin (Cbl) when bound by each of HC, IF and TC is in the compact unfolded structure, where the surface of Cbl is protected to a large degree by the proteins wrapping around the Cbl.. Each of HC, IF and TC are quite promiscuous in binding to Cbl and will each bind cyanocobalamin, hydroxocobalamin, adenosylcobalamin, methylcobalamin or Co(II)cobalamin. HC will also bind cobamide, which is not bound either by IF or TC.
Once Holo TC is bound by cellular receptors, the Vitamin B12-bound by TC is taken up by various cells in the body via Receptor-Mediated Endocytosis. Once inside the cell the Cbl-TC dissociates and the Cbl is transferred as one of the co-enzyme forms methylCo(III)B12 to methionine synthase in the cytoplasm and AdenosylCo(III)B12 to methylmalonylCoA mutase in the mitochondria, respectively (Matthews et al, 2007; Evans et al, 2000). During the binding to the two enzymes both AdenosylCo(III)B12 and MethylCo(III)B12 assume an extended shape, wherein the imidazole ring dissociates from the central cobalt atom, thus allowing greater reactivity of the central cobalt atom.
.
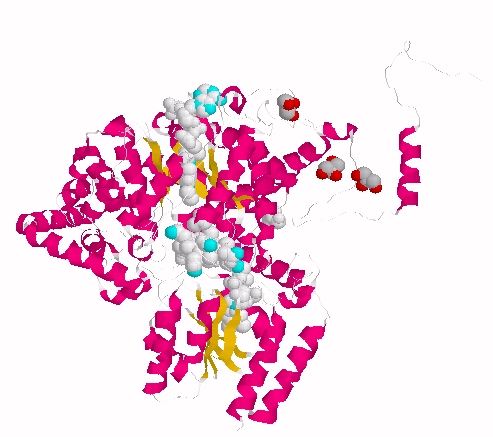
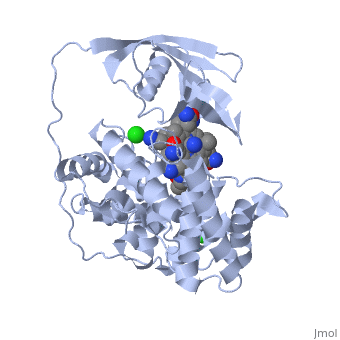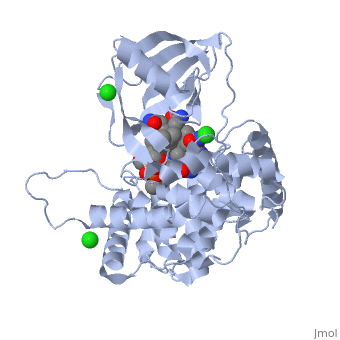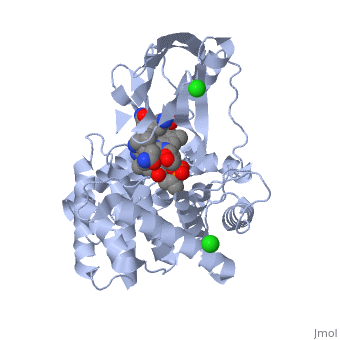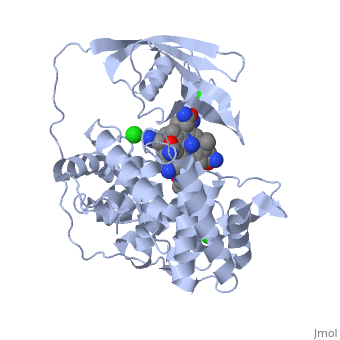
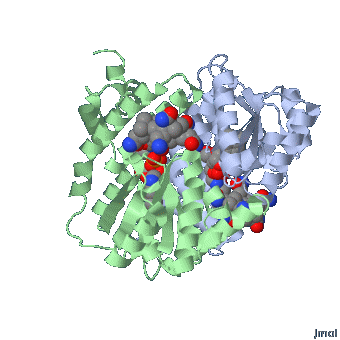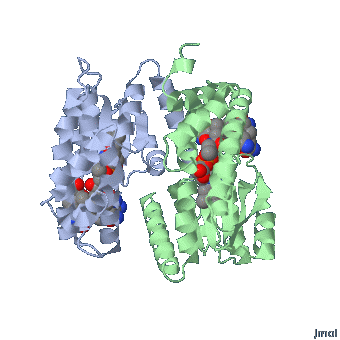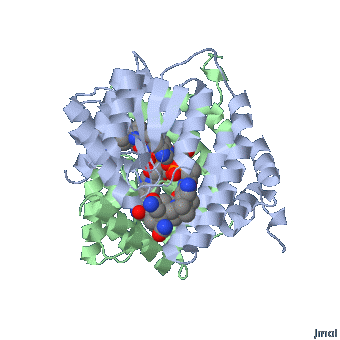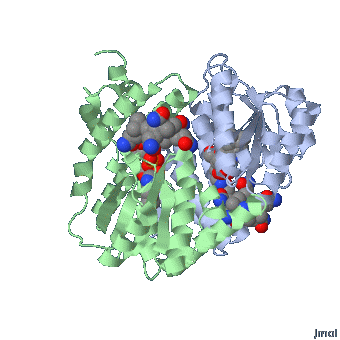
Extended form of AdenosylCbl (Left) MMACoA-bound AdoCbl (Right)
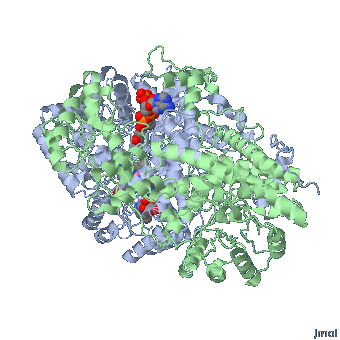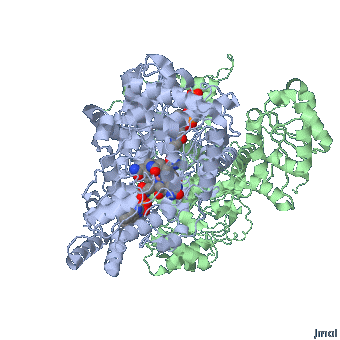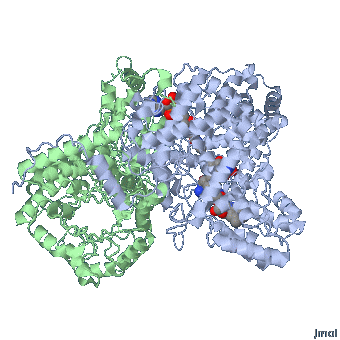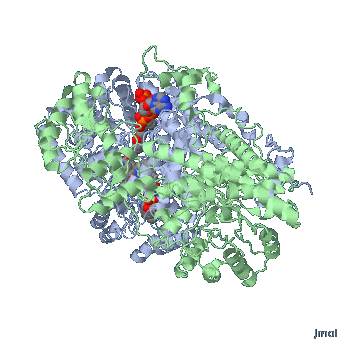
MMACoA bound AdoCbl
Mancia et al, 1996
MethylCbl in extended form (Left) and
Methionine Synthase bound MeCbl (Right)
Methionine synthase + MeCbl
Free vitamin B12 when injected into the body, or delivered transdermally, has three potential fates. It can bind to either ApoTC, or ApoHC or is rapidly cleared from the circulation by the kidney. in normal tissue, 50%-70% of intracellular Cbl is Adenosyl B12 with only 5% to 15% present as methyl B12, with the rest either inactive Co(II)B12 or hydroxyB12 (Collins et al, 1999). In the circulation, The situation is slightly different in the brain, with methylcobalamin being the major form found (Zhang et al, 2016). Vitamin B12 that is not excreted gradually accumulates in the liver, with gradual accumulation also being seen in the salivary glands, where presumably it is bound to haptocorrin. Potentially this would mean that vitamin B12 is recycled via secretion of haptocorrin-bound Cbl into the stomach and re-uptake of IF-bound Cbl in the small intestine. Such recycling would not occur in individuals who have PA, or those who are taking ant-acids or PPIs. In the circulation there are four vitamin B12 binding proteins, apo-transcobalamin - Transcobalamin bound a VB12 binding free Haptocorrin (HC)(Apohaptocorrin) - VB12 - bound to Haptocorrin (Holohaptocorrin). When vitamin B12 is Injected, the free/holo transcobalamin and holocobalamin binding sites are rapidly saturated, and then excess vitamin B12 is lost to urine. This effectively means that when a person is very deficient in vitamin B12 it can be very hard for them to become replete unless they have repeated injections. Orally is even harder as the maximum uptake is 3-6 ug per feed. (see Sokoloff et al, 1952)
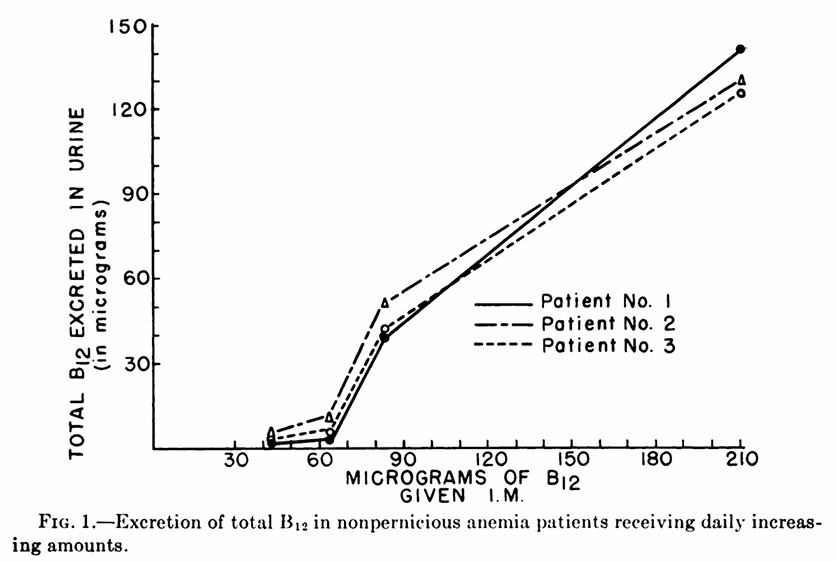
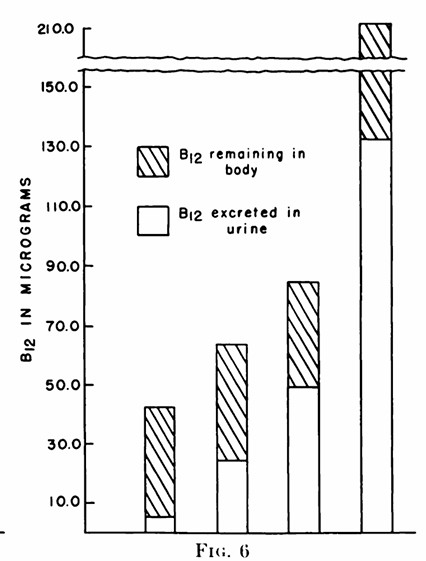
Injection of 42, 84 and 211 ug of vitamin B12 into patients, and collection in urine, to determine the excreted amount of vitaminB12
Oral administration of radiolabelled vitamin B12 shows that there is delayed appearance of vitamin B12 in the blood, liver, skin and muscle, which drops off within 24 hours. Very little material goes to the lungs, or spleen. The majority of the 10 ng dose passes down the intestine and is eventually lost in faeces. Calculations suggest less than 50% of the maximal absorptive dose reaches the circulation. Increasing the dose can increase the amount absorbed non-specifically to a level of 0.1 to1% of the dose administered.
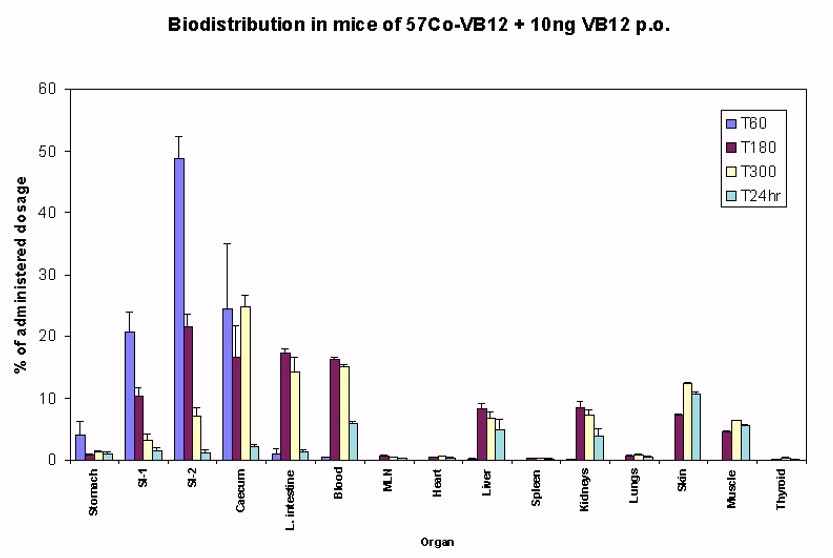
Russell-Jones et al, 1997
Brain-loading with vitamin B12 appears to happen in the foetus, with up to 17% of transplacentally obtained B12 tracking to the brain. Brain accumulation accelerates with foetal life and hence babies that are premature are likely to have lower levels of B12 in their brains at birth. Thereafter there is a gradual drop in brain-B12 over the ensuing years, and it appears as if there is very little replenishment of the brain with B12 as we age (Zhang et al, 2016). Studies looking at imaging with radioactive B12 show very, very little accumulation in brains (Collins et al, 1999).
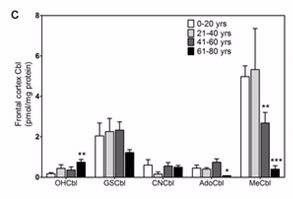
Zhang et al, 2016
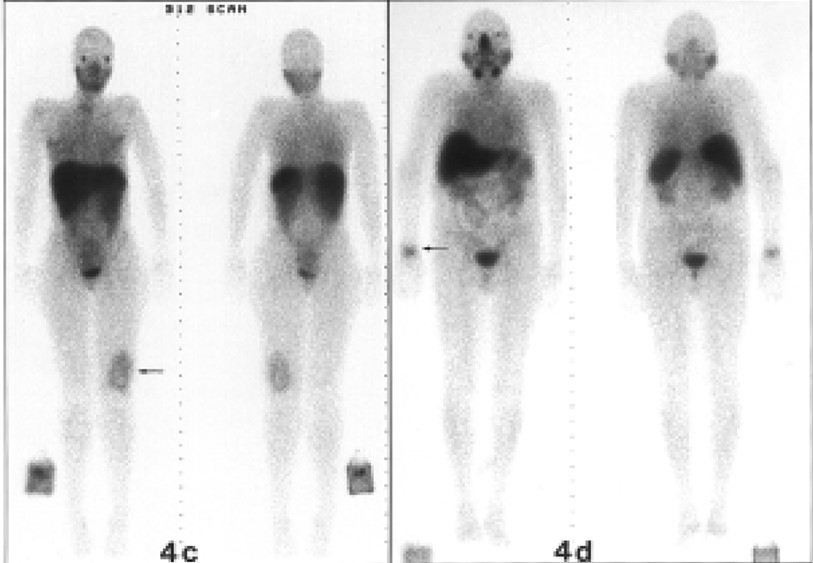
Collins et al, 1999
Lack of uptake into the brain of radiolabelled vitamin B12. Note the accumulation of label in the salivary glands, liver, kidneys and bladder
Alpers DH, Russell-Jones G Gastric intrinsic factor: the gastric and small intestinal stage of cobalamin absorption. a person journey. Bioechimie 2013 95: 989-94
Alsenz, Russell-Jones, Westwood et al. Oral absorption of peptides through the cobalamin (vitamin B12) pathway in the rat intestine Pharm Res 2000 17: 825-832
Furger, et al. Structural basis for universal corrinoid recognition by the cobalamin transport protein haptocorrin. J.Biol.Chem. 2003 288: 25466-25476
Gordon et al. Expression of functional intrinsic factor using recombinant baculovirus. Methods Enzymol 1997 281: 255-61
Wen et al. Functional expression in Pichia pastoris of human and rat intrinsic factor. Biochim Biophys Acta 2000 1490: 43-53 PMID 107856616
Matthews et al Crystal structure of human intrinsic factor: cobalamin complex at 2.6-A resolution. PNAS 2008 104: 17311-6 PMID 17954916
Evans et al Protection of radical intermediates at the active site of adenosylcobalamin-dependent methylmalonyl-CoA mutase Biochem 2000 39: 9213-21PMID 10924114
Collins et al Tumor imaging via Indium 111-labeled DTPA-adenosylcobalamin. Mayo Clin Proc 1999 74: 687-691
Mancia et al. Methylmalonyl-CoA mutase. Structure 1996 4: 339-350
Zhang et al Decreased brain levels of vitamin B12 in aging, autism and schizophrenia Plos One 2016
SOKOLOFF MF, SANNEMAN EH Jr, BEARD MF. Urinary excretion of vitamin B12. Blood. 1952 Feb;7(2):243-50. PMID: 14886418.
.
Copyright © 2018
vitaminb12deficiency.info. All Rights Reserved.
The statements on this site compose a compendium of generally recognized signs
of vitamin B12 deficiency, and problems that can then ensue They also are formulated from a summary of relevant
scientific publications. In addition they may contain some forward looking
statements of a general nature.
Reproduction in whole or in part in any form or medium without express written
permission is prohibited