Vitamin B12 (cobalamin), the only cobalt containing vitamin, is also the vitamin that is present in least amount in the body. Despite this, a deficiency in vitamin B12 can have huge consequences both in obtaining energy from food and for processing energy within the energy chain, as well as through its involvement in over 200 methylation reactions in the body..
Intracellular vitamin B12 functions as a co-factor for only two enzymes in the body.
AdenosylCo(III)B12 (adenosylcobalamin) is a co-factor for methylmalonylCoA mutase in the mitochondria and the other co-enzyme form is methylCo(III)B12 (methylcobalamin, which acts as a co-factor for methionine synthase in the cytoplasm (Matthews et al, 2007; Evans et al, 2000). During the binding to the two enzymes both AdenosylCo(III)B12 and MethylCo(III)B12 assume an extended shape, wherein the imidazole ring dissociates from the central cobalt atom, thus allowing greater reactivity of the central cobalt atom.
.
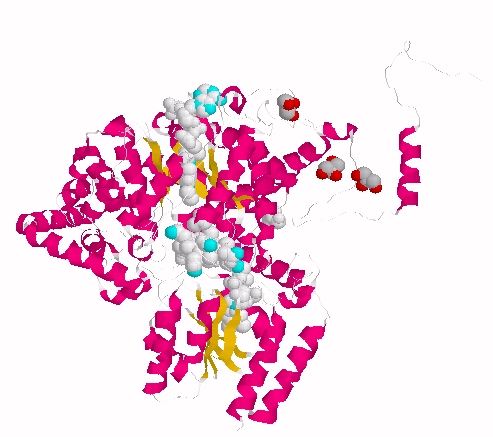
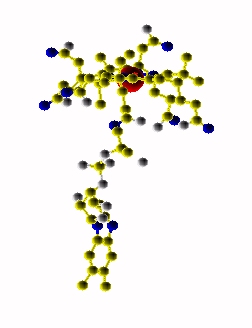
Extended form of AdenosylCbl (Left) MMACoA-bound AdoCbl (Right)
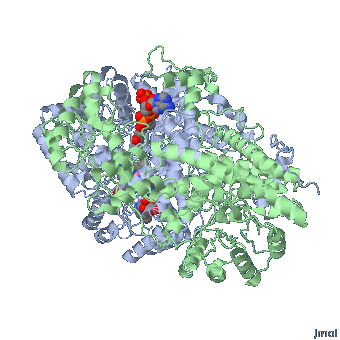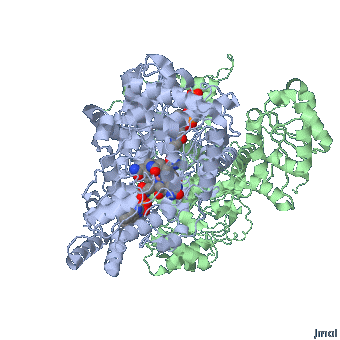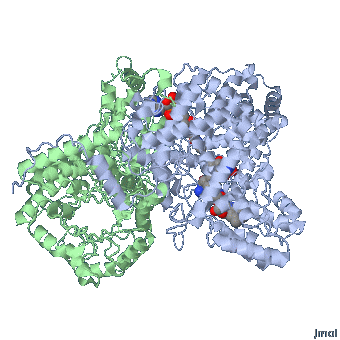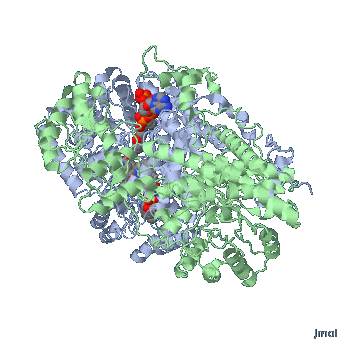
MMACoA bound AdoCbl
Mancia et al, 1996
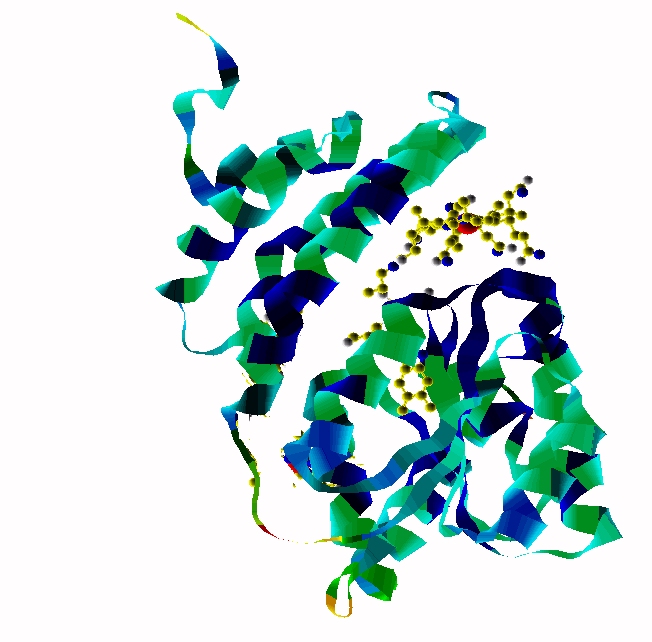
MethylCbl in extended form (Left) and
Methionine Synthase bound MeCbl (Right)
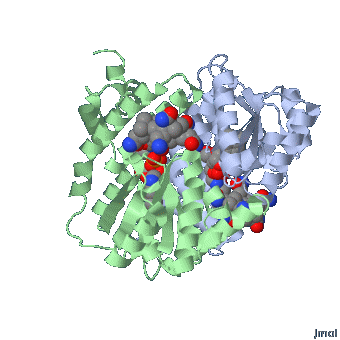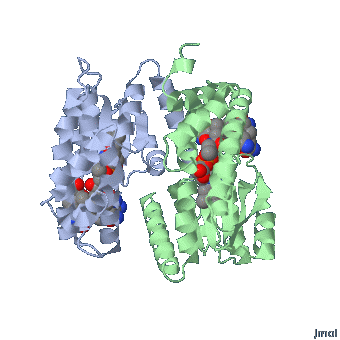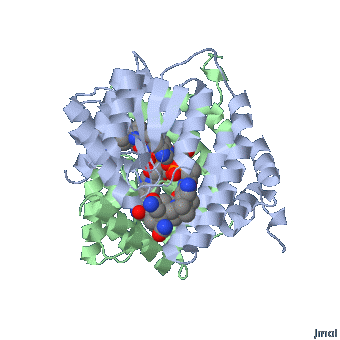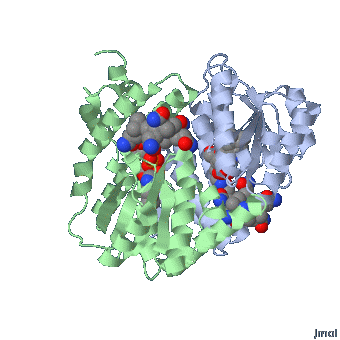
Methionine synthase + MeCbl
Approximately 50%-70% of intracellular Cbl is Adenosyl B12 with only 5% to 15% present as methyl B12, with the rest either inactive Co(II)B12 or hydroxyB12 (Collins et al, 1999). MMACoA-mutase is involved in obtaining energy from branched chain amino acids such as Isoleucine, valine, threonine, thymine and also from odd-chain fatty acids. Lack of activity of the enzymes results in an increase in levels of MMA. MMA is neurotoxic, which is a problem in the brain as this dicarboxylic acid is poorly transported across the blood-brain barrier, leading to trapping within the CNS, which in turn leads to neurological debilities.
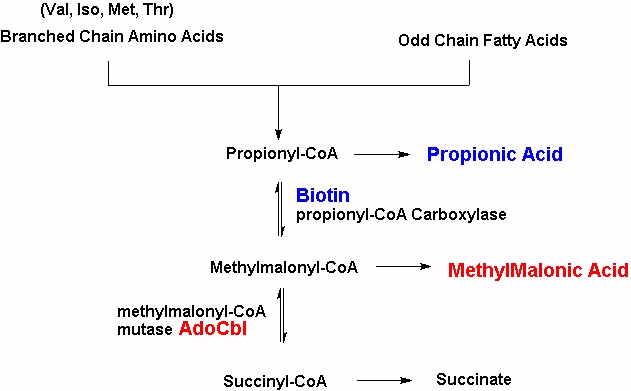
MMACoA mutase activity
Methionine synthase (MTR/MS) with its co-factor MethylCo(III)B12 plays an essential role in recycling methionine to enable the ultimate synthesis of the methylating agent, s-Adenosylmethionine (SAM, or SAMe). In this reaction, MS transfers the methyl group from MethylCo(III)B12 to homocysteine, yielding Co(I)B12 and methionine. Subsequently the cofactor is remethylated using the methyl group from 5-methyltetrahydrofolate. Thus, cycling of the enzyme is critically dependent upon the presence of incoming 5MTHF. In the absence of 5MTHF, the Co(I)B12 is oxidized to inactive Co(II)B12.
Cycling of MethylCo(III)B12 is essential for the generation of more and more SAM from dietary methionine. In methylCo(III)B12 deficiency homocysteine becomes elevated and SAM levels drop, with an accompanying drop in methylation. In this case there is also an accompanying decrease in the movement of homocysteine through to cystathionine, which is the precursor for the production of hydrogen sulphide and also for the production of free sulfur for synthesis of iron-sulphur complexes, which are essential several enzymes including lipoate synthase, aconitase, succinate dehydrogenase (complex II), Complex I, and GABA-aminotransferase. Potentially deficiency in methylCo(III)B12 would effect around 200 methylation reactions in the body.
There are approximately 200 methylation reactions that are potentially affected by a deficiency in methyl B12, including the production of adrenalin and melatonin, inactivation of histamine and the neuroamines, dopamine, nor-adrenalin, adrenalin and melatonin, production of CoQ10, carnitine, creatine, methylation of DNA and histones, structural modifications of lysine, histidine and arginine, Methylation of myelin basic protein, inactivation of metals, such as arsenic, and many others. Lack of methylation has been implicated in the development of autoimmune diseases such as systemic lupus erythematosis, rheumatoid arthritis, systemic sclerosis, and Sjogren's syndrome. Lack of methylation has been implicated in conditions such as adrenal fatigue.
Melatonin has often been considered the sleep hormone. Production of melatonin depends upon a methylation reaction, and so in methylB12 deficiency, there can be a lack of production of melatonin in the pineal gland, and can result in an alteration in sleep patterns with characteristic insomnia, with around 30% of people over the age of 50 yr exhibiting sleep problems. Melatonin, though has a much greater role in the body than just controlling the circadian rhythm and melatonin has been shown to act as an analgesic, an anxiolytic agent, and has been used in the treatment of fibromylagia and neuropathic pain. Melatonin has also been shown to have a role in the management of depression, epilepsy, Alzheimer's disease, diabetes, obesity, alopecia, migraine, some cancers and in immune and cardiac disorders. A deficiency in production of melatonin has been linked to CNS disorders such as stroke, OCD and shizophrenia (Karu and Shyu, 2018).
Melatonin has also been shown to stimulate neurogenesis and is involved in the differentiation of neuronal stem cells to form oligodendrocytes and Schwann cells, and as such is essential for myelination of nerves.
Melatonin was originally thought to only be synthesized in the brain, but a considerable amount of melatonin is produced in the gut, where it is essential for maturation of the gut. Lack of synthesis of gut melatonin, due to vitamin B12 deficiency, has been associated with IBS.
Production of Adrenalin depends upon a methylation reaction in which nor-adrenalin is methylated by the enzyme Phenylethanolamine-N-methyltransferase. One of the earliest signs of vitamin B12 deficiency is orthostatic hypotension, initially described in 1962 (Kalbfliesch and Woods, 1962), and infrequently thereafter (White etal, 1981; Eisenhofer etal, 1982; Johnson 1987; Lossos and Argov, 1991; Girard etal, 1998; Beitzke etal 2002; Moore etal, 2004; Ganjehei etal, 2012), however, the association between methyl B12 and the formation of adrenalin does not seem to have been commonly known until much later, despite the existence of the enzyme PNMT in the adrenals being known as early as 1968 (Laduron and Belpaire, 1968; Kitabche and Williams, 1969).
Deficiency of vitamin B12 has been shown to cause mitochondrial disease, and as such potentially is implicated in the known mitochondrial diseases, including autism, cerebral palsy, Parkinson's disease, epilepsy, dementia, Alzheimer's disease, Huntington's disease, developmental delay, Chronic Fatigue, Fibromyalgia, Lou Gehrig's, Atypical Learning disorder, muscular dystrophy and diabetes.
Evans et al Protection of radical intermediates at the active site of adenosylcobalamin-dependent methylmalonyl-CoA mutase Biochem 2000 39: 9213-21PMID 10924114
Collins et al Tumor imaging via Indium 111-labeled DTPA-adenosylcobalamin. Mayo Clin Proc 1999 74: 687-691
Mancia et al. Methylmalonyl-CoA mutase. Structure 1996 4: 339-350
Zhang et al Decreased brain levels of vitamin B12 in aging, autism and schizophrenia Plos One 2016
Kaur T and Shyu B-C Melatonin: a new-generation therapy for reducing chronic pain and improving sleep disorder-related pain. Adv Exp Med Biol 2018 1099:229-251
Kalbfliesch and Woods Orthostatic hypotension associated with pernicious anemia.... JAMA 1962 182:198-200
White etal Pernicious anemia seen initially as orthostatic hypotension. Arch Intern Med 1981 141:1543-4
Eisenhofer etal Deficient catecholamine release as the basis of orthostatic hypotension in pernicious anemia. J Neurol Neurosurg Psychiatry 1982 45:1053-5
Johnson GE Reversible orthostatic hypotension of pernicious anemia. JAMA 1987 257:1084-6
Lossos and Argov Orthostatic hypotension induced by vitamin B12 deficiency. J Am Geriatr Soc 1991 38:601-2
Girard etal Orthostatic hypotension revealing vitamin B12 deficiency. Rev Neurol 1998 154:324-4
Laduron and Belpaire Biochem Pharmacol 1968 17:1127-40
Kitabchi and Williams BBA 1969 178:181-4
Beitzke etal Autonomic dysfunction and hemodynamics in vitamin B12 deficiency. Auton Neurosci. 2002 97:45-54
Moore etal Orthostatic tolerance in older patients with vitamin B12 deficiency before and after vitamin B12 replacement. Clin Auton Res 2004 14:67-71
Ganjehei etal Orthostatic hypotension as a manifestation of vitamin B12 deficiency. Tex Heart Inst. J 2012 39:722-3
.
Copyright © 2018
vitaminb12deficiency.info. All Rights Reserved.
The statements on this site compose a compendium of generally recognized signs
of vitamin B12 deficiency, and problems that can then ensue They also are formulated from a summary of relevant
scientific publications. In addition they may contain some forward looking
statements of a general nature.
Reproduction in whole or in part in any form or medium without express written
permission is prohibited